HORIZON EIC NaV1.5-CARED: Julien Barc (2023 – 2028)
Julien Barc is the coordinator of the HORIZON EIC grant entitled NaV1.5 regulation fine-tuning as a therapy for cardiac Conduction and Arrhythmic diseases at Risk of suddEn Death
Abstract
The voltage-gated sodium channel (Na-channel; NaV1.5) is a central component of cardiac electrogenesis. Its dysfunction can lead to ventricular fibrillation and sudden cardiac death. While NaV1.5 represents a highly relevant therapeutic target for prevention of life- threatening cardiac arrhythmias, therapies that target the expression or function of this channel are non-existent. Such a therapy would provide for the first time an alternative for the invasive and costly cardioverter defibrillator or pacemaker therapies currently used to treat patients suffering from a Brugada syndrome and/or a progressive cardiac conduction disease.
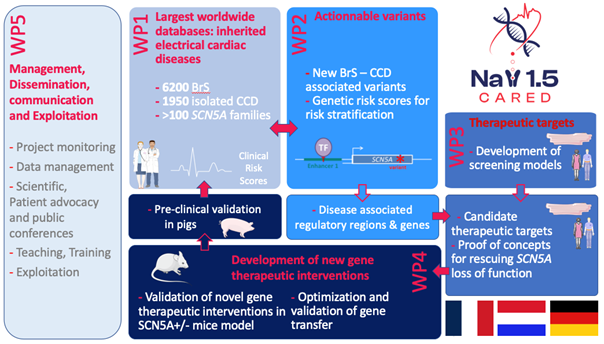
NaV1.5-CARED consortium proposes to capitalize on their largest worldwide cohorts of patients with inherited cardiac electrical and conduction defects to uncover regulatory regions and proteins that modulate NaV1.5 expression and function with the goal to develop and validate innovative therapies to restore the function of NaV1.5.
Objectives
We have three objectives: 1/ predict the risk of (fatal) arrhythmia and conduction defect at the individual level by developing personalized polygenic risk score (PRS), 2/ characterize the molecular mechanism associating regulatory DNA regions and genes with the cardiac diseases to uncover new therapeutic targets, 3/ and develop new candidates for therapeutic intervention able to restore Nav1.5 loss of function.
Work plan
After databases harmonization, we will conduct whole genome genetic studies to identify variants associated with ventricular arrhythmia and conduction defects and generate PRS relevant to stratify the risk of arrhythmia and degree of conduction defect. Genetic studies will also be used to identify new therapies that will be evaluated in dedicated and high-throughput human cardiomyocytes derived induced pluripotent cell models and then through pre-clinical trials, performed on mouse and pig models.
Principal collaborators
AMIN Ahmad Shoaib, BEZZINA Connie, Experimental Cardiology Department of Amsterdam UMC
CHRISTOFFELS Vincent, Department of Medical Biology of Amsterdam UMC
GOURRAUD Jean-Baptiste, CHU Nantes
CORNEC Marine, GenoA, Nantes
CHARPENTIER Flavien, DE WAARD Michel, l’institut du thorax, Team II, Nantes
DIECKE Sebastian, MDC Berlin
BOINK Gerard, PacingCure, Amsterdam
BEROUD Rémy, Smartox, Grenoble
Project website
https://www.nav15-cared.eu